Could we be using bird models to guide some of our own dinner table disputes over who will get the last slice of bread? In a study published this month by the Journal of Evolutionary Biology, researchers from the University of Lausanne in Switzerland suggested that hungry barn owl nestlings (Tyto alba) might be using vocal signals while parents are off hunting, in order to negotiate who will get the next incoming meal.
We know that bird songs can help monogamous couples identify their partners and recognize their young and that these songs are often influenced by a combination of genetics and environmental factors, such as learning. But there is still a lot we don’t know about the mechanisms by which individuals recognize and remember calls of their siblings. Previous research has shown that barn owl nestlings have a sibling-sibling communication system that allows them to cooperate in the distribution of food. But how is the fairness of food distribution ensured? Who is keeping track?
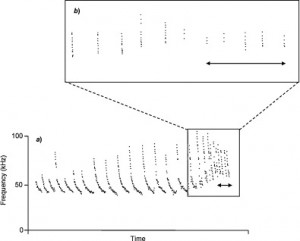
The way the sib-sib communication system works is that when the nestlings signal to each other, the nestling that is the most vocal or loudest or vocalizes at the highest frequency is granted priority to the incoming prey without having to compete with it’s siblings. The more food deprived a nestling is, the louder it will compete. The researchers, here, predicted that the signals of barn owl nestlings are individualized and therefore able to be recognized and remembered by their siblings. They suspect that the ability to recognize the individuality of sibling signals might have evolved as a way to strengthen the honesty of the calls. But if the nestlings are all hungry, how is it that the bigger nestlings, which are naturally louder, don’t beat out the smaller ones each time?
This question gets even more complicated when you take into account the fact that barn owls are asynchronous hatchers: during the breeding season, barn owl moms lay an egg every 2-3 days and the eggs hatch in the order that they are laid so that the oldest and youngest nestlings can be up to a few weeks apart in age. It seems intuitive that the older nestlings, then, would win every vocal competition—but they don’t. In fact, according to what the researchers found, the nestlings actively avoid simultaneous signaling, implying that all siblings want to hear the calls of their nest-mates so that they can recognize their identity and calibrate their own calls in order to compete fairly. The older siblings have been shown to produce fewer negotiation calls when competing with juniors in the nest, seemingly in order to level the playing field. The individual recognition system also ensures that if a nestling cheats, he or she will be punished by the siblings, who will steal the food away.
The bottom line here is that not only do these signals help the nestlings convey their hunger, but the signals have evolved to serve to identify siblings and ensure that the hungriest sibling wins the prize. To learn more, take a look at the original paper in this month’s issue of the Journal of Evolutionary Biology.
Reference: Dreiss, A. N., C.A Ruppli, and A. Roulin. 2014. Individual vocal signatures in barn owl nestlings: does individual recognition have an adaptive role in sibling vocal competition? Journal of Evolutionary Biology 27: 63-75.