Chitin and chitosan have a large number of commercial applications due to unusual and often unstudied properties of both compounds. Both are found in many commonly used products as well as products being tested for consumer use. They will probably come to be found in more products soon due to the eco-friendliness of the chitin structure. Not only is it biodegradable and renewable, it is also digestible and safe for human and animal ingestion. It is estimated that 100 billion tons of chitin is produced biologically and degraded every year.
In medicine, chitin and chitosan have been shown to improve the immune systems in plants and animals and preventing bacterial and viral infections. In rabbits with chitosan feed additives, the production of helpful intestinal bacteria increased and prevented microbial infections. It is also proven that chitin increases cell reproduction in animal and human wounds, decreasing healing time. It is now an active ingredient in many bandages and topical wound creams.
Chitin and chitosan have also been shown to moisturize human skin, prevent visible aging and protect hair from mechanical damage. Eco-friendly cosmetic products and textiles feature chitin and its derivatives as active ingredients.
Chitosan also reacts with certain chemicals in food and water. In polluted water, chitosan will react with polyanionic polymers and metal ions to precipitate polyelectrolyte and chelate complexes. It also absorbs radioisotopes. This allows for the clarification of drinking water and the recovery of proteins and metal ions from industrial waste water. A layer of chitosan also prevents the release of carbon dioxide and ethyelene from fruits and vegetables, helping them stay fresh longer.
By using the unusual properties of the chitin molecule, it is possible to develop innovative ways to improve and produce commercial products with the exoskeleton of a crustacean, something that would ordinarily be considered waste.
Category: Chitin
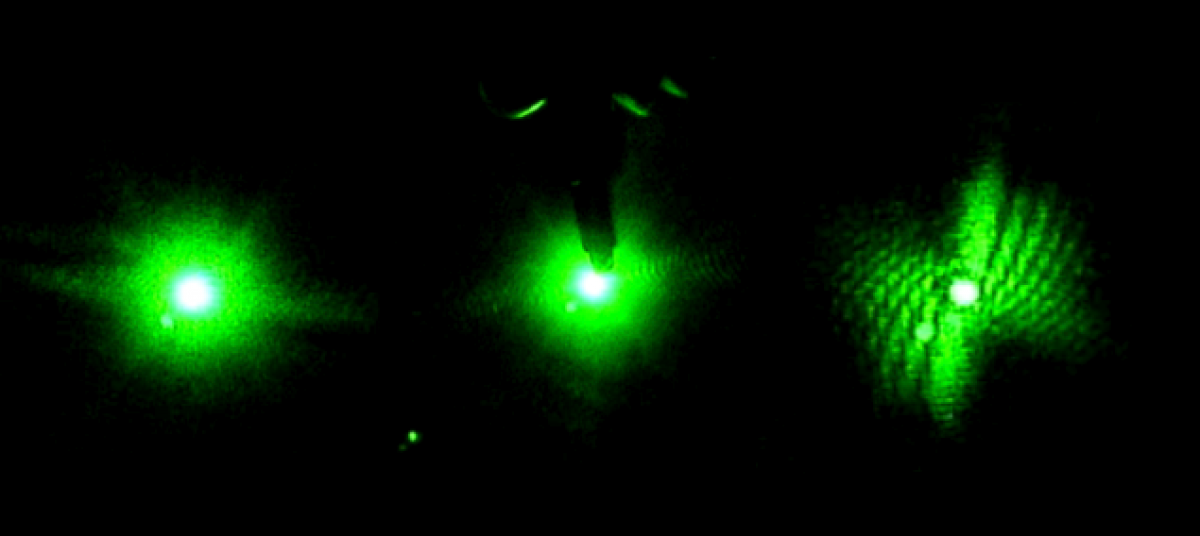
Information about the optical properties of chitin.
Chitosan: a Derivative of Chitin
The most commercially useful derivative of chitin is chitosan. It is made by the deacetylation of the pure chitin compound. Typically, pure chitin is harvested from shrimp, crab and lobster shells disposed of by restaurants. In this process, an acetyl group (molecular formula C2H3O+) is removed from a single chitin molecule [10]. This changes the physical and chemical properties of the molecule, introducing a more restricted number of orientations for chitosan chains than for chitin chains. While still forming orthorhombic micelles, these micelles have a smaller depth and length (8.9 Å depth and 17.0 Å length) than α-chitin micelles but have the same width. Pure chitosan is structurally weaker than that of pure chitan. It begins decomposing at 184°C, and is soluble in dilute acids [14]. It is still, however insoluble in many common solvents such as concentrated acid, alcohol and acetone. Despite it’s weaker constitution, chitosan is much more common in commercial and industrial products and has a wider variety of uses, ranging from medical to agricultural.
Basic Chemical Structure
Chitin was discovered in 1811 by Henri Braconnot, a chemist who worked mostly with plants and fungi. He originally named it fungine, because it was a main structural component in fungi cell walls. In 1823, chitin was found in insects as well and its name was changed. Chitin bears a close resemblance to the plant structure component cellulose. In fact, the two are close to identical except for the small amount of nitrogen in chitin [6]. The theoretical value of nitrogen in chitin is about 6.85% while experimental results range from 6 to 7% depending on the extraction method used [14].
Pure chitin is a high molecular weight polymer that is formed by anhydro-N-acetylglucosamine residues joined together. These molecules can form very long chains, up to several hundred residues or between 0.1 and 1 microns long. The chains do not branch and are highly ordered [3].
When chitin is in its purest form, it is insoluble in most common solvents, making it an excellent material for the exoskeletons of arthropods. Chitin is also colorless in its macromolecule state, but pigmentation and structural color have been observed in naturally occurring chitin.
Long chains of chitin generally bond together to form larger groups called micelles or crystallites. These micelles have a rhombic shape and can also associate into larger chains called microfibers. Microfibers are the most common macroformation of biological chitin but film structure chitin has also been observed. It is unclear how the chitin chains are ordered in a film structure, but they display interesting optical properties including light interference.
X-ray diffraction experiments show two main kinds of micelle structures, which have been named α- and β-chitin. In both, the height and depth of the unit micelle is the same (9.4 Å depth, 10.4 Å height [14]) but the length of the micelles differ (19.25 Å in α-chitin, 22.15 Å in β-chitin [14].) The form most commonly found in beetles is the α form. There is also a third form, the γ-chitin form, but it is much less common than the first two.
References
[1] Argyros, A., S. Manos, M.C. J. Large, D.R. McKenzie, G.C. Cox, and D.M Dwarte. “Electron Tomography and Computer Visualisation of a Three-dimensional ‘photonic’ Crystal in a Butterfly Wing-scale.” Micron 33.5 (2002): 483-87.
[2] Biro, L.P., K. Kertesz, Z. Vertesy, G. Mark, Zs. Balint, V. Lousse, and J.P. Vigneron. “Living Photonic Crystals: Butterfly Scales – Nanostructure and Optical Properties.” Materials Science and Engineering C 27.5-8 (2007): 941-46.
[3] Carlstrom, Diego. “The Crystal Structure Of α-Chitin (Poly-N-Acetyle-D-Glucosamine).” Journal of Cell Biology 3.5 (1957): 669-83.
[4] “The Chitin Crystallite in Arthropod Cuticle.” Journal of Cellular Science 21 (1876): 73-81.
[5] Galusha, Jeremy W., Lauren R. Richey, Matthew R. Jorgensen, John S. Gardner, and Michael H. Bartle. “Study of Natural Photonic Crystals in Beetle Scales and Their Conversion into Inorganic Structures Via a Sol–gel Bio-templating Route.” Journal of Materials Chemistry 20.7 (2010): 1277-284.
[6] Goosen, Mattheus F. A. Applications of Chitin and Chitosan. Lancaster, PA: Technomic Pub., 1997.
[7] Joannopoulos, J. D., Pierre R. Villeneuve, and Shanhui Fan. “Photonic Crystals: Putting a New Twist on Light.” Nature 386.6621 (1997): 143-49.
[8] Kinoshita, Shuichi, Shinya Yoshioka, and Kenji Kawagoe. “Mechanisms of Structural Colour in the Morpho Butterfly: Cooperation of Regularity and Irregularity in an Iridescent Scale.” Proceedings of the Royal Society B: Biological Sciences 269.1499 (2002): 1417-421.
[9] Marsano, E., G. Conio, R. Martino, A. Turturro, and E. Bianchi. “Fibers Based on Cellulose-chitin Blends – Marsano – 2001 – Journal of Applied Polymer Science.” Wiley Online Library. 18 Dec. 2001. Web. .
[10] Mogilevskaya, E. L., T. A. Akopova, A. N. Zelenetskii, and A. N. Ozerin. “The Crystal Structure of Chitin and Chitosan.” Polymer Science Series A 48.2 (2006): 116-23.
[11] Neville, A. C., and S. Caveney. “Scarabaeid Beetle Exocuticle As An Optical Analogue Of Cholesteric Liquid Crystals.” Biological Reviews 44.4 (1969): 531-62.
[12] Parker, A. R. “The Diversity and Implications of Animal Structural Colours.” Journal of Experimental Biology 201.16 (1998): 2343-347.
[13] Rao, Vijayalakshmi, and A.T. Ramaprasad. “Optical and Conductivity Studies of Pseudo-doped Chitin-polyaniline Blend.” Journal of Applied Polymer Science 106.1 (2007): 309-13. 14 June 2007.
[14] Richards, Albert G. “Glucosamine, N-Acetylglucosamine, Chitobiose, and Chitin.” Integument of Arthropods. St. Paul: Univeristy of Minnesota, 1951. 9-25.
[15] Seago, Ainsley E., Parrish Brady, Jean-Pol Vigneron, and Tom D. Schultz. “Gold Bugs and Beyond: a Review of Iridescence and Structural Colour Mechanisms in Beetles (Coleoptera).” Journal of The Royal Society Interface 6.SUPPL. 2 (2009): S165-184.
[16] Stoddart, P. R., P. J. Cadusch, T. M. Boyce, R. M. Erasmus, and J. D. Comins. “Optical Properties of Chitin: Surface-enhanced Raman Scattering Substrates Based on Antireflection Structures on Cicada Wings.” Nanotechnology 17.3 (2006): 680-86.
[17] Vukusic, P., J.R. Sambles, C.R. Lawrence, and R.J. Wootton. “Structural Colour. Now You See It–now You Don’t.” Nature 410.6824 (2001): 36.
[18] Vukusic, Pete, and J. Roy Sambles. “Photonic Structures in Biology.” Nature 424.6950 (2003): 852-55.
[19] Yablonovitch, E. “Photonic Band-gap Structures.” Journal of the Optical Society of America B 10.2 (1993): 283-95.
[20] Yablonovitch, Eli. “Inhibited Spontaneous Emission in Solid-State Physics and Electronics.” Physical Review Letters 58.20 (1987): 2059-062.
Optical Properties of Chitin
In order to further study the optical properties of beetles, I must study the structure and optical properties of chitin.
In order to further understand the optical properties of beetles, I must first learn about the structure and optical properties of chitin.
Chitin is a polymer that is derived from glucose. It is similar to keratin, the protein in human fingernails, and cellulose. Chitin is most commonly found in the shells of arthropods such as crustaceans and insects. It can also be found in the cell walls of fungi and in mollusks.
A derivative of chitin, chitosan, has been found to have healing and blood clotting properties when used on human wounds. In addition, chitosan is used in agriculture to promote plant immune systems and as strengthening and binding agents in many commercial products.
Though it has many industrial and medicinal uses, chitin in its natural forms exhibits some interesting optical properties. The study of these properties will increase understanding of the material and improve its usefulness to humans.